|
|
| Untersuchte Arbeit: Seite: 16, Zeilen: 1 ff. (entire page) |
Quelle: Anézo 2003 Seite(n): 30, 31, Zeilen: 30:8ff; 31:1ff |
|---|---|
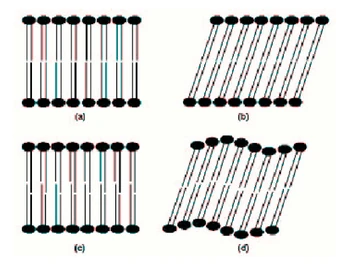
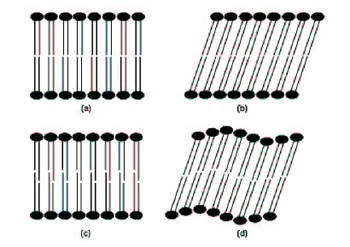
Figure 1.3. Gel phases of phospholipids: (a): Lβ untilted phase; (b): Lβ’ tilted phase; (c): LβI interdiginated [sic] phase; (d): Pβ’ rippled phase. Fluid phases Lamellar liquid crystalline phase Lα. Upon heating a gel phase, the phospholipid chains melt to a liquid-like conformation, transforming usually to the lamellar Lα phase. The rate of lateral diffusion of the lipids increases by at least two orders of magnitude and the hydrocarbon chains exhibit a high tendency to isomerize, alternating conformations about the carbon-carbon bonds. In this disoriented fluid state, phospholipid movements are still inhibited by some organizational constraints for instance the phospholipid molecules cannot translate extensively in the direction normal to the bilayers surface but remain rather confined within the bilayers, in accordance with the hydrophobic effect. The phospholipids are therefore neither in a true lipid state, in which their movement would be isotropic, nor in a solid state, but in a so-called liquid crystalline state. A liquid crystal retains at least one dimension of order relative to the [solid state.] |
[page 30]
Figure 1.9: Gel phases of phospholipids: (a): Lβ untilted phase; (b): Lβ' tilted phase; (c): LβI interdigitated phase; (d): Pβ' rippled phase. 1.2.2.3 Fluid phases Lamellar liquid crystalline phase Lα Upon heating a gel phase, the phospholipid chains melt to a liquid-like conformation, transforming usually to the lamellar Lα phase. The rate of lateral diffusion of the lipids increases by at least two orders of magnitude and the hydrocarbon chains exhibit a high tendency to isomerize, alternating conformations about the carbon-carbon bonds. In this disordered fluid state, phospholipid movements are still inhibited by some organizational constraints: for instance, the phospholipid molecules cannot translate extensively in the direction normal to the bilayer surface but remain rather confined within the bilayer, in accordance with the hydrophobic effect. The phospholipids are therefore neither in a true liquid state, in which their movement would be isotropic, nor in a solid [page 31] state, but in a so-called liquid crystalline state. A liquid crystal retains at least one dimension of order relative to the solid state |
The source is not given |
|